Abstract
Background: Three drug combinations are the standard treatment for newly diagnosed multiple myeloma (NDMM). However, induction, consolidation, and maintenance therapy have not been standardized in Japan. Therefore, in this single arm Phase II study, we evaluated bortezomib-based induction, autologous stem cell transplantation (ASCT), bortezomib-based consolidation, and bortezomib maintenance in transplant eligible NDMM patients and assessed clinical outcomes as well as the minimal residual disease (MRD) status.
Methods: Patients received four cycles of CyBorD induction therapy with bortezomib 1.3 mg/m2 and cyclophosphamide 300 mg/m2 on day 1, 8, 15, and 22 and dexamethasone 40 mg on day 1-3, 8-10, 15-17, and 22-24 for the first two cycles and day 1, 8, 15, and 22 for the last two cycles of four 28-day cycles. Peripheral blood stem cells were collected after cyclophosphamide 2 g/m2 for 2 days, which was followed by melphalan 200 mg/m2 and ASCT. Three months after ASCT, patients received consolidation treatment with three cycles of CyBorD identical to the last two cycles of the induction therapy followed by maintenance therapy with bortezomib 1.3 mg/m2 on day 1 and 15 of a 28-day cycle, for 24 months. The primary end-point was the complete response (CR) rate after consolidation therapy and assessed as an interim analysis. Responses were assessed according to the International Myeloma Working Group (IMWG) criteria. MRD was assessed using an allele-specific oligonucleotide real-time quantitative polymerase chain reaction (qPCR). The toxicity was evaluated according to the National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (CTCAE).
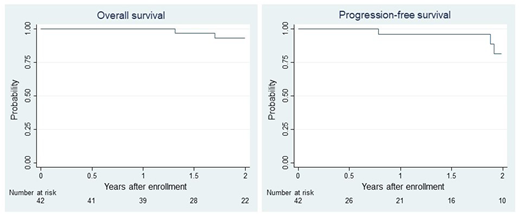
Results: From August 2013 to May 2016, 42 (25 male and 17 female) patients with a median age of 58 (range 42-65) years with NDMM were found eligible and enrolled in 15 centers in Japan. The International Staging System (ISS) values were 1 in 17 (40%), 2 in 20 (48%), and 3 in 5 patients (12%). Adverse cytogenetics of 17p deletion, t(4;14), and t(14;16) were observed in 20%, 18%, and 3% of the evaluable patients, respectively. Following four induction cycles of CyBorD, the overall response rate (ORR) was obtained in 71% of patients, including a CR/sCR of 10% and very good partial response (VGPR) of 26%. One of the evaluable 11 patients showed MRD negativity after induction therapy. Four patients discontinued the protocol during the induction therapy because of grade 4 interstitial pneumonia (n = 2), prolonged grade 3 drug eruption (n = 1), and grade 1 delirium (n = 1). Four patients discontinued the protocol due to doctor judgement (inadequate efficacy, n = 2; repetitive infection, n = 1; grade 3 neutropenia, n = 1). A total of 26 of the 42 patients completed ASCT following the protocol and 18 achieved VGPR/CR, including CR in 10 patients. Three of the evaluable 10 patients showed MRD negativity after ASCT. The 2-year overall and progression-free survival rates were 93% (95% confidence interval [CI], 76%-98%) and 81% (95% CI, 51%-94%), respectively (Figure 1).
Conclusions: CyBorD with ASCT for NDMM resulted in relatively high CR rates in the investigated Japanese population, although a relatively high incidence of discontinuation of therapy was observed. Dose and schedule modification of induction therapy may be necessary in Japanese populations. Clinical trial information: UMIN000010542.
Imada:Celgene: Honoraria; Bristol-Meyers Squibb: Honoraria; Takeda: Honoraria; Novartis: Honoraria; Nippon Shinyaku: Honoraria; MSD: Honoraria; Chugai: Honoraria; Sumitomo Dainippon Pharma: Honoraria; Kyowa Hakko Kirin: Honoraria; Shire Japan: Honoraria; Ono: Honoraria; Mundipharma: Honoraria. Takaori-Kondo:Bristol-Myers Squibb: Honoraria; Pfizer: Honoraria; Janssen Pharmaceuticals: Honoraria; Celgene: Honoraria, Research Funding; Novartis: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.